More Information
Submitted: March 26, 2026 | Accepted: May 08, 2026 | Published: May 11, 2026
Citation: Oriba DL, Catherine N, Nampogo AM, Anyango MW, Bagasha P. Intra-Abdominal Hypertension Complicating Acute Pancreatitis with a Protracted Course of Severe COVID-19 Infection: A Case Report. J Clin Intensive Care Med. 2026; 11(1): 6-10. Available from:
https://dx.doi.org/10.29328/journal.jcicm.1001057
DOI: 10.29328/journal.jcicm.1001057
Copyright license: © 2026 Oriba DL, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Intra-abdominal Hypertension (IAH); Acute Pancreatitis (AP); COVID-19; Acute Kidney Injury (AKI); Hemodialysis; Enterocutaneous fistula
Intra-Abdominal Hypertension Complicating Acute Pancreatitis with a Protracted Course of Severe COVID-19 Infection: A Case Report
Dan Langoya Oriba1,2* , Nagadya Catherine3, Adrian Mwota Nampogo1, Mbadi Winnie Anyango1 and Peace Bagasha1
, Nagadya Catherine3, Adrian Mwota Nampogo1, Mbadi Winnie Anyango1 and Peace Bagasha1
1Department of Medicine, School of Medicine, Makerere University College of Health Sciences, Division of Nephrology, Kiruddu National Referral Hospital, Kampala, Uganda
2Department of Internal Medicine, St. Mary’s Hospital Lacor, Gulu, Uganda
3Department of Surgery, School of Medicine, Makerere University College of Health Sciences, Mulago National Referral and Specialized Hospital, Kampala, Uganda
*Corresponding author: Oriba Dan Langoya, MBchB, Department of Medicine, School of Medicine, Makerere University, Upper Mulago Hill Road, P.O. Box 7072, Kampala, Uganda, |Email: [email protected]
Introduction: The presence of intra-abdominal hypertension is common but often underappreciated in patients with severe acute pancreatitis, partly related to the effects of the inflammatory process, causing retroperitoneal edema, fluid collections, ascites, and ileus, and partly resulting from medical interventions, especially aggressive fluid resuscitation. It seems to be an early phenomenon and is associated in some patients with early multiple organ failure, like acute kidney injury, but it can also manifest at a later stage, often associated with local pancreatic complications.
Case: In this case report, we detailed the management of a 30-year-old male who presented with acute pancreatitis complicated by severe intra-abdominal hypertension requiring surgical intervention and a hospital course complicated by severe COVID-19 pneumonia at the height of the COVID-19 pandemic in Uganda.
Conclusion: We reported a case of AP leading to IAH, which was clinically diagnosed by a high index of suspicion, increasing abdominal distension with absent bowel sounds, and reduced urine output in a resource-limited setting where standard bladder measurement of IAH was not possible. The hospital course was protracted by the development of a hospital-acquired severe COVID-19 pneumonia as well as surgical complications of a burst abdomen and an enterocutaneous fistula. A multidisciplinary team approach, including medical and surgical teams as well as the patient’s family, was pivotal in ensuring a good patient outcome in a resource-constrained setting.
Intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS) are established causes of morbidity and mortality in critically ill patients [1]. The incidence of intra-abdominal hypertension (IAH) is high, approximately 60% - 80% patients with severe acute pancreatitis [2], and a high prevalence of IAH has recently been estimated at between 32% and 65% in both medical and surgical intensive care units [3,4]. Etiologically, the two most common causes of acute pancreatitis are cholelithiasis/choledocholithiasis and alcohol [5]. Because IAH is clearly related to organ dysfunction in acute pancreatitis, and because it is reversible and to some extent preventable, IAH should definitely be considered a therapeutic target. Nonsurgical measures to reduce IAP should be considered first, and when ineffective, surgical approaches may be necessary in patients with persistent organ dysfunction [2].
We present the case of a 30-year-old male patient of concern who came to Kiruddu National Referral Hospital emergency room as a self-referral with a history of progressive abdominal distention with severe colicky pain for 4 days, which was generalized and radiating to the back. It was associated with high-grade fevers. He also reported a history of non-projectile vomiting, with no hematemesis, constipation, or jaundice. Significantly, before referral, there was reduced urine output with volume estimated to be less than 500 mL in 24 hours and notably associated with generalized body swelling and facial puffiness.
Socially, history was notable for alcohol intake with a CAGE Score of 2/4; however, quantification of units was not possible due to different beverages taken. Important negatives included no history of cough, chest pain, or easy fatigability and a negative HIV serology.
On examination at the emergency department, the patient was sick-looking with anasarca and febrile with an axillary temperature of 38.6 degrees. There was no pallor of mucous membranes or jaundice, capillary refill was normal (< 2s), Glasgow Coma Scale (GCS) was normal (15/15), tachycardia of 106 beats per minute, and had a normal blood pressure (120/73mmHg). The abdomen was distended and tense with generalized rebound tenderness marked at the epigastrium, and bowel sounds were absent.
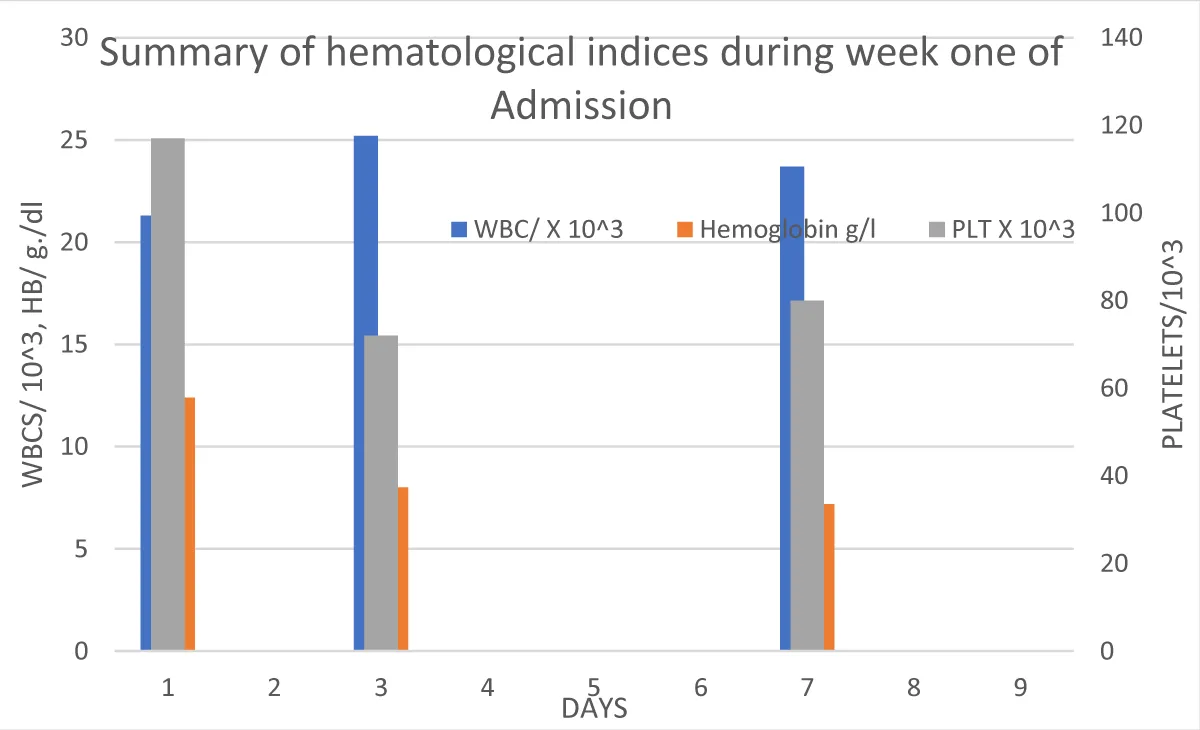
Laboratory investigations showed a leukocytosis, predominantly neutrophils, and thrombocytopenia (summarized in figure one). Admission renal functions test and electrolytes showed a markedly elevated Creatinine at 1808.7 μmol/l, and reduced urine output of 110 mls in 24 hours (< 0.3 mls/kg in 24 hours) corresponding to Stage 3 Acute Kidney Injury by Kidney Injury Network (AKIN) classification, complicated by mild hyperkalemia (serum potassium 5.51 mEq/L). Lastly, serum urea was 39 mmol/L; the calculated BUN/creatinine ratio was less than 10.
In addition, the patient demonstrated persistent high-grade fever, marked neutrophilic leukocytosis, thrombocytopenia, and progressive organ dysfunction suggestive of severe sepsis. Blood culture and other microbiological investigations could not be performed due to resource limitations during the peak of the COVID-19 pandemic. However, the clinical diagnosis of sepsis was supported by systemic inflammatory response features together with acute kidney injury and hemodynamic deterioration.
Liver chemistry was significant for mild hypoalbuminemia (28.7 g/l), an elevated Gamma-Glutamyl Transpeptidase (GGT) (133 u/l), and other liver enzymes were normal. Pancreatic enzymes showed an elevated serum lipase (445 U/L) while serum amylase was mildly elevated (161.5 U/L), indicating acute alcoholic pancreatitis. Supine abdominal x-ray only showed mild right pleural effusion, and abdominal ultrasound scan revealed ascites with features of paralytic ileus. The diagnosis was Intra-abdominal Hypertension secondary to Acute Pancreatitis with complications of severe sepsis, Acute Kidney Injury (AKI), AKIN stage 3 with electrolyte imbalance, thrombocytopenia, and paralytic ileus.
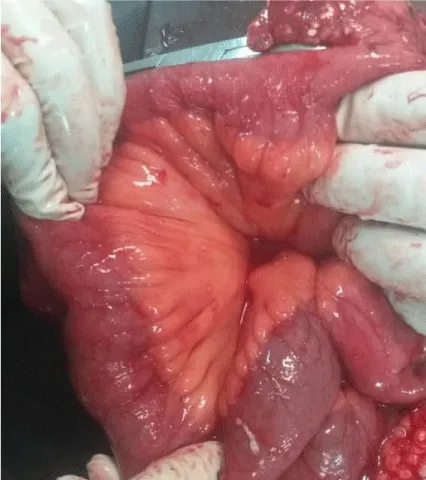
Management plan included abdominal decompression with nasogastric tube placement, intravenous fluids, broad-spectrum intravenous antibiotics, surgical review, and hemodialysis. Empirical antibiotic therapy was initiated because of suspected severe sepsis in the setting of acute pancreatitis with systemic inflammatory response and worsening organ dysfunction. Antibiotic selection was guided by local treatment protocols and clinical response. Close monitoring of urine output, renal function, electrolyte balance, and hemodynamic status was performed throughout admission. The decision to perform a decompressive abdominal laparotomy was made, and found to have ischemic and edematous bowel with minimal peritoneal fluid. Postoperatively, hemodialysis was initiated on day one. Urine output and serum creatinine were monitored, and hemodialysis was suspended when urine output was greater than 1200 mls. The patient developed a high-output enterocutaneous fistula (ECF), which was managed conservatively with diet, rehydration, care for the skin, and antibiotics (Figures 1-3).
Figure 1: Ischemic and edematous bowel with minimal peritoneal fluid.
Figure 2: Summary of hematological Parameters (Full blood count) during week one of admission.
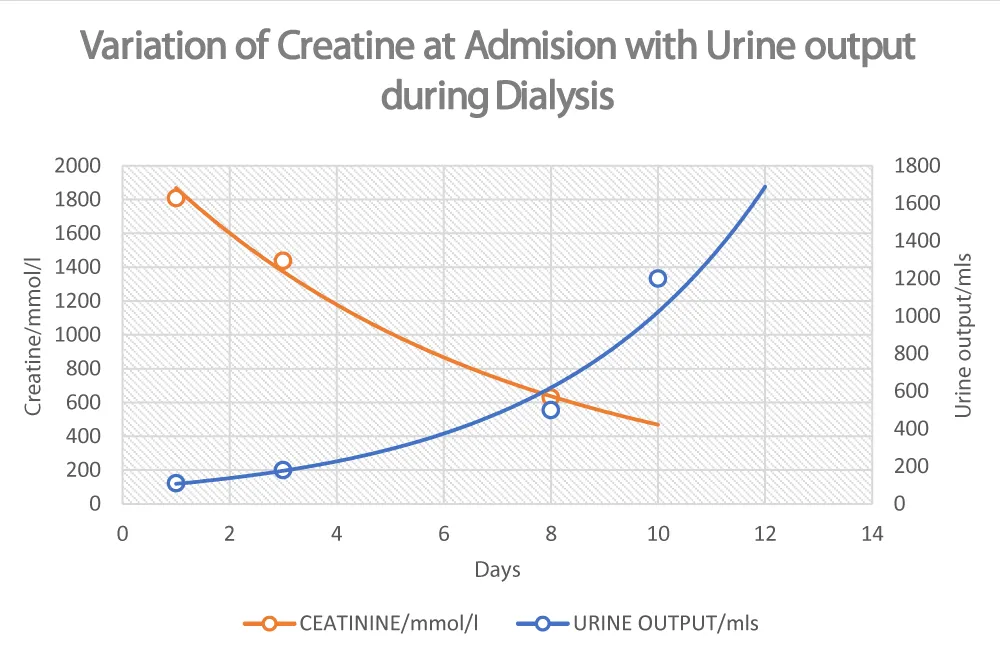
Figure 3: Variation of serum creatinine and urine output during dialysis with days of hospital stay.
Hemodialysis was suspended on day 10 when urine output was 1200 mls in 24 hours, i.e., 0.9 mls/kg/hr, with corresponding serum creatinine of 500 Μmol/l. Patient continued to show remarkable improvement with a reduction in the flow of the Enterocutaneous fistula, and renal function was normal by day 16.
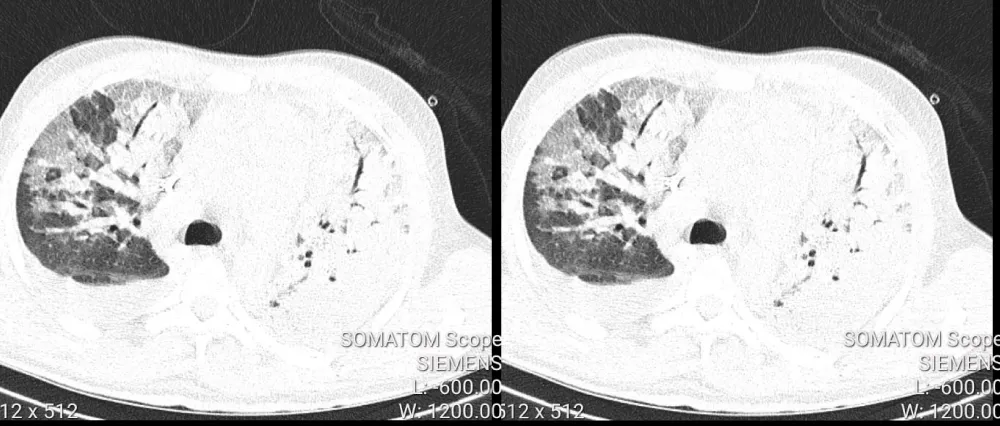
However, on day 24 postoperatively in the ward, the patient developed a new onset of cough, high-grade fevers, and progressive worsening difficulty in breathing. He was tachycardic at 114 bpm, and tachypneic at 36 breaths per minute with a SpO₂ OF 70% at room air. SARS-CoV-2 Rapid antigen was negative, and PCR yielded positive results. Chest CT-scan showed relatively dense ground glass infiltration and consolidation diffusely affecting both lungs with air bronchogram, and when PCR later became positive following transfer to the COVID-19 treatment unit of Mulago, a score of CORAD 6/6 was then given (Figure 4).
Figure 4: High resolution chest CT-scan (HRCT) showed relatively dense ground glass infiltration and consolidation diffusely affecting both lungs with air bronchogram.
A diagnosis of Severe COVID-19 was made. The patient was not tolerating supplemental oxygen (Non-rebreather mask 20 L/min). We then considered using high flow nasal cannula (HFNC) with a positive end expiratory pressure (PEEP) of 4 mmHg, responsible for recruiting and maintaining alveoli open. Close monitoring for worsening vital signs & work of breathing,s and our target SpO2 was at 92% - 96%, we achieved an improvement in SP02 from 70% at room air to 75% on NRM, then to 80% - 91% on HFNC. We did not institute noninvasive ventilation (NIV) and prone positioning due to the tight balance with positioning in ECF. Other management included intravenous antibiotics, dexamethasone, and optimizing fluid intake and output to offset losses at the enterocutaneous fistula site. The patient recovered and was transferred to the surgical ward after 21 days of treatment, where Secondary wound closure was performed. He was discharged home after 42 days of hospitalization and continues to maintain a good general condition.
This specific case emphasizes the utility of a multidisciplinary approach, in resource-limited settings, in the management of the critically ill patient and an unprecedented road to recovery at the height of the COVID-19 pandemic. We highlight a difficult case of the patient with AP complicated by IAH, leading to multiple organ dysfunction with AKI and Paralytic ileus, surgical decompression of IAH complicated by the development of a burst abdomen with an enterocutaneous fistula, and finally, hospital-acquired severe COVID-19 pneumonia. In the present case, we diagnosed IAH clinically based on a high index of suspicion, increasing abdominal distension, and reduced urine output.
The diagnosis of intra-abdominal hypertension in this patient was primarily based on progressive clinical deterioration and characteristic examination findings in the absence of standard bladder pressure measurements. Clinical features supporting the diagnosis included progressive tense abdominal distension, generalized abdominal tenderness, absent bowel sounds suggestive of paralytic ileus, worsening oliguria progressing to severe acute kidney injury, respiratory compromise, and generalized edema. Furthermore, the patient demonstrated significant clinical improvement following decompressive laparotomy and supportive management, further supporting the diagnosis of clinically significant intra-abdominal hypertension. This case highlights the diagnostic challenges encountered in resource-limited settings where invasive intra-abdominal pressure monitoring tools are often unavailable.
While appreciating the absence of IAP measurement to guide diagnosis and decision making.
Intra-abdominal hypertension (IAH) and its deleterious effects are present in at least one-third of critically ill patients. Increased recognition of IAH has led to a significant reduction in the incidence of abdominal compartment syndrome (ACS) [6]. IAH can be expected in at least 17% of patients presenting with a diagnosis of AP to a typical tertiary care hospital (prevalence increasing to 50% in those with severe disease) [7]. The optimal treatment of choice for patients with IAH/ACS should take into account three critical elements: (1) the measured IAP value (or the degree/magnitude of IAP increase); (2) organ dysfunction characteristics (or the impact of increased IAP); and (3) the nature and course of the underlying disease [8].
Insights into the diagnosis and management of AP acute pancreatitis are evolving, with many treatment strategies that were once considered the standard of care eventually being discarded as non- beneficial or even harmful. Both medical treatment and surgery have advanced significantly, but morbidity and mortality of severe acute pancreatitis have remained high, and the course of the disease is often protracted, especially in resource-limited settings where diagnostic capability and management options may be limited [2].
Several mechanisms appear to increase the risk of IAH in patients with AP, ranging from the disease pathophysiology to the treatment given. They include: intra-abdominal volume increases due to pancreatic and peri-pancreatic edema, often fueled by aggressive fluid resuscitation, initiated to compensate for central hypovolemia due to third spacing. Paralytic ileus and reduced abdominal wall compliance due to edema and pain are also major contributors seen in this case.
The decision to manage this patient surgically was justified by refractoriness of IAH to medical management, which included decompression of intraluminal contents with nasogastric tube insertion and fluid removal with ultrafiltration on hemodialysis. Refractoriness was evidenced by worsening ileus and an increasingly tense abdominal distension and progression into septic shock. Animal studies have found that pancreatic perfusion is decreased in IAH [9], which may further increase the risk of pancreatic necrosis and bacterial translocation, the presumed pathway for pancreatic and systemic infection [2].
There are still a lot of gaps and controversies in the optimal management of patients with acute pancreatitis and IAH using decompressive laparotomy. In 2005, De Waele, et al. [10], reported universal mortality following decompressive laparotomy for ACS in four patients and cautioned against its routine use. Subsequently, a large study by Chen, et al. [11] in 2008 demonstrated obvious amelioration in clinical variables within 24 h after decompression for ACS in patients with severe AP, and 25% patients survived. They then recommended a low threshold for intervention in patients with AP determined by the presence of IAH and early signs of changes in physiologic variables. Furthermore, the retrospective study in 26 consecutive cases of decompressive laparotomy for ACS in severe AP by Mentula, et al. [12] in 2010 showed that decompressive laparotomy improved renal and respiratory function when surgical decompression was carried out within the first four days after disease onset. This prompted the conclusion that early decompression reduced mortality. However, we notice a caveat with the complication of high enterocutaneous fistulae and severe COVID-19 pneumonia, which, combined, highly increase morbidity and mortality from complicating management.
Emerging evidence suggests that COVID-19 infection may contribute to pancreatic injury and worsening systemic inflammation through multiple mechanisms, including cytokine-mediated injury, endothelial dysfunction, microvascular thrombosis, and dysregulated immune responses. In patients with severe acute pancreatitis, the coexistence of COVID-19 may amplify the inflammatory cascade and increase the risk of multiorgan dysfunction, prolonged hospitalization, and secondary infectious complications. In the present case, the development of severe COVID-19 pneumonia likely contributed to the protracted clinical course and further complicated fluid management, respiratory support, and nutritional optimization. These observations are consistent with previous reports describing poorer outcomes among critically ill patients with concurrent severe COVID-19 infection and acute pancreatitis.
The hospital-acquired severe COVID-19 pneumonia management was complicated. Challenges in this case included: 1) the need for judicious rehydration to be balanced with already compromised respiratory functions, 2) lifesaving maneuvers like prone position couldn’t be done, 3)a complex balance of electrolytes in already constrained COVID-19 treatment unit (insufficient resources to monitor electrolytes more than once every 48 hours), 4) monitoring nutritional/daily calorie needs in the setting of an high flow enterocutaneous fistula, 5) treatment of sepsis and care of a burst abdomen while avoiding steroid therapy. This required a multidisciplinary team, spanning from social workers, psychologists, intensive care physicians, nurses, and surgeons, to optimize care and ensure a good outcome. The patient’s family was equally pivotal to good patient outcomes by providing psychosocial support as well as timely financial support to buy medicines and pay for laboratory investigations not available in the hospital.
A limitation in this case was the inability to obtain comprehensive microbiological investigations, including blood culture testing, due to resource constraints during the COVID-19 pandemic period. Consequently, the diagnosis of sepsis was primarily clinical and supported by available laboratory findings and organ dysfunction parameters. Despite these limitations, the patient’s clinical presentation and response to multidisciplinary management strongly supported the working diagnosis and treatment approach.
We reported a case of AP leading to IAH diagnosed clinically by a high index of suspicion, increasing abdominal distension with absent bowel sounds, and reduced urine output in a resource-limited setting where standard bladder measurement of IAH was not possible. The hospital course was protracted by the development of hospital-acquired severe COVID-19 pneumonia as well as surgical complications of a burst abdomen and an enterocutaneous fistula. A multidisciplinary team approach, including medical and surgical teams as well as the patient’s family, was pivotal in ensuring a good patient outcome in a resource-constrained setting.
Consent: The patient provided informed consent to publish their case details and any accompanying images.
Special thanks go to the staff of Kiruddu National Referral Hospital Division of Nephrology and Surgery, and to the staff of Mulago Hospital, a specialized and Referral Hospital, National COVID-19 treatment Unit.
- Malbrain MLNG, Cheatham ML, Kirkpatrick A, et al. Results from the International Conference of Experts on Intra-abdominal Hypertension and Abdominal Compartment Syndrome. I. Definitions. Intensive Care Med. 2006;32(11):1722-1732. Available from: https://doi.org/10.1007/s00134-006-0349-5
- De Waele JJ, Leppäniemi AK. Intra-abdominal hypertension in acute pancreatitis. World J Surg. 2009;33(6). Available from: https://doi.org/10.1007/s00268-009-9994-5
- Malbrain MLNG, Deeren D, De Potter TJR. Intra-abdominal hypertension in the critically ill: it is time to pay attention. Curr Opin Crit Care. 2005;11(2). Available from: https://journals.lww.com/co-criticalcare/Fulltext/2005/04000/Intra_abdominal_hypertension_in_the_critically.11.aspx .
- Sugrue M. Abdominal compartment syndrome. Curr Opin Crit Care. 2005;11(4). Available from: https://journals.lww.com/co-criticalcare/Fulltext/2005/08000/Abdominal_compartment_syndrome.8.aspx .
- Garber A, Frakes C, Arora Z, Chahal P. Mechanisms and Management of Acute Pancreatitis. Girotra M, ed. Gastroenterol Res Pract. 2018;2018:6218798. Available from: https://doi.org/10.1155/2018/6218798
- Kimball EJ. Intra-abdominal hypertension and abdominal compartment syndrome: a current review. Curr Opin Crit Care. 2021;27(2). Available from: https://journals.lww.com/co-criticalcare/Fulltext/2021/04000/Intra_abdominal_hypertension_and_abdominal.14.aspx .
- Jaipuria J, Bhandari V, Chawla AS, Singh M. Intra-abdominal pressure: Time ripe to revise management guidelines of acute pancreatitis? World J Gastrointest Pathophysiol. 2016;7(1):186-198. Available from: https://doi.org/10.4291/wjgp.v7.i1.186
- De Laet IE, Malbrain MLNG, De Waele JJ. A Clinician’s Guide to Management of Intra-abdominal Hypertension and Abdominal Compartment Syndrome in Critically Ill Patients. Crit Care. 2020;24(1):97. Available from: https://doi.org/10.1186/s13054-020-2782-1
- Endo K, Sasaki T, Sata N, et al. Elevation of intra-abdominal pressure by pneumoperitoneum decreases pancreatic perfusion in an in vivo porcine model. Surg Laparosc Endosc Percutaneous Tech. 2014;24(3). Available from: https://doi.org/10.1097/SLE.0b013e3182937bd6
- De Waele JJ, Hoste E, Blot SI, Decruyenaere J, Colardyn F. Intra-abdominal hypertension in patients with severe acute pancreatitis. Crit Care. 2005;9(4):R452. Available from: https://doi.org/10.1186/cc3754
- Chen H, Li F, Sun J-B, Jia J-G. Abdominal compartment syndrome in patients with severe acute pancreatitis in the early stage. World J Gastroenterol. 2008;14(22):3541-3548. Available from: https://doi.org/10.3748/wjg.14.3541
- Mentula P, Hienonen P, Kemppainen E, Puolakkainen P, Leppäniemi A. Surgical Decompression for Abdominal Compartment Syndrome in Severe Acute Pancreatitis. Arch Surg. 2010;145(8):764-769. Available from: https://doi.org/10.1001/archsurg.2010.132